Content
The content of medical knowledge in this section of the site of the Lactology Foundation is intended for the practical needs of doctors, pharmacists and students in these specialties. It is more than reasonable to consult other authoritative medical sources before using our medical knowledge.
- Lung in Health and Disease
- General Approach to Patients
With Respiratory Disorders - Evaluating Lung Structure and Function
- Interstitial Lung Diseases
- Pulmonary Vascular Diseases
- Disorders of the Pleura,
Mediastinum, and Chest Wall - Respiratory Failure
- Lung Transplantation
- Perioperative Pulmonary Management
- COVID-19 Pulmonary Management
- Congenital Lung Malformations
- Sleep-Related Disorders
Pleural Effusion
A pleural effusion is a collection of fluid abnormally present in the pleural space, usually resulting from excess fluid production and/or decreased lymphatic absorption. It is the most common manifestation of pleural disease, and its etiologies range in spectrum from cardiopulmonary disorders and/or systemic inflammatory conditions to malignancy. Approximately 1.5 million pleural effusions are diagnosed in the United States each year.
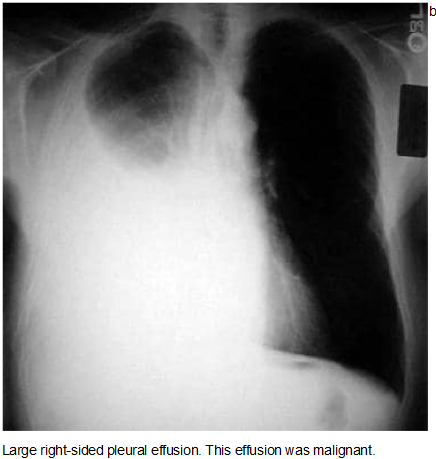
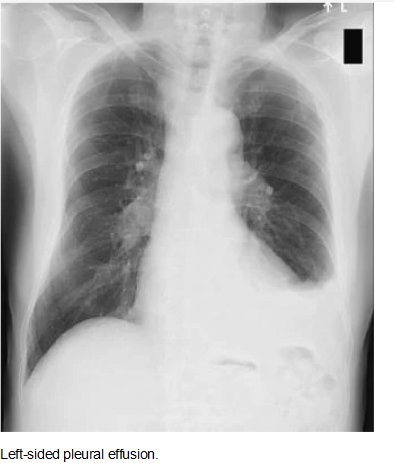
Anatomy
The pleural space (cavity) in a
healthy patient is a potential space sandwiched
between the parietal and visceral pleurae. The
parietal pleura completely lines the inner chest
wall surface of the thoracic cavity, inclusive
of the bilateral medial mediastinum, the
subcostal left and right diaphragmatic leaflets,
and the innermost muscle surface of the ribs and
associated musculature. The visceral pleura
tightly envelops both lungs completely, folding
into the interlobar fissures, meeting the
parietal pleura at the hilar root of the lungs.
The right and left pleural cavities are
separated in healthy people by the anterior and
posterior mediastinum.
Playing a vital role
in respiration, the potential space of the
pleural cavity in healthy patients joins the
natural outward movement of the chest wall to
that of the natural inward movement of the lungs
via two mechanisms. First, the potential space's
relative vacuum sustains the visceral and
parietal pleurae's extreme adherence and is
uninterrupted and not disrupted. Second, a
diminutive volume of pleural fluid (calculated
at 0.13 mL/kg of body weight under normal
situations) serves as the lubricant to
facilitate the normal physiological sliding
motion of both pleural surfaces against each
other during inspiration and expiration. This
small volume of lubricating fluid is maintained
via a delicate balance of hydrostatic and
oncotic pressure and peripheral sulcal lymphatic
drainage; disturbances in any of these
mechanisms may lead to pathology and, possibly,
manifest as a pleural effusion.
Etiology
The normal pleural space contains
approximately 10 mL of fluid, representing the
balance between (1) hydrostatic and oncotic
forces in the visceral and parietal pleural
capillaries and (2) persistent sulcal lymphatic
drainage. Pleural effusions may result from
disruption of this natural balance.
Presence
of a pleural effusion heralds an underlying
disease process that may be pulmonary or
nonpulmonary in origin and, furthermore, that
may be acute or chronic. Although the etiologic
spectrum of pleural effusion can be extensive,
most pleural effusions are caused by congestive
heart failure, pneumonia, malignancy, or
pulmonary embolism.
Deferent mechanisms may play a role in the
formation of pleural effusion:
Iterated
permeability of the pleural membranes (eg,
inflammation, malignancy, pulmonary embolism)
• Reduction in intravascular oncotic pressure
(eg, hypoalbuminemia due to nephrotic syndrome
or cirrhosis)
• Increased capillary
permeability or vascular disruption (eg, trauma,
malignancy, inflammation, infection, pulmonary
infarction, drug hypersensitivity, uremia,
pancreatitis)
• Increased capillary
hydrostatic pressure in the systemic and/or
pulmonary circulation (eg, congestive heart
failure, superior vena cava syndrome)
•
Reduction of pressure in the pleural space (ie,
due to an inability of the lung to fully expand
during inspiration); this is known as "trapped
lung" (eg, extensive atelectasis due to an
obstructed bronchus or contraction from fibrosis
leading to restrictive pulmonary physiology)
• Decreased lymphatic drainage or complete
lymphatic vessel blockage, including thoracic
duct obstruction or rupture (eg, malignancy,
trauma)
• Increased peritoneal fluid with
microperforated extravasation across the
diaphragm via lymphatics or microstructural
diaphragmatic defects (eg, hepatic hydrothorax,
cirrhosis, peritoneal dialysis)
• Movement of
fluid from pulmonary edema across the visceral
pleura
• Persistent increase in pleural fluid
oncotic pressure from an existing pleural
effusion, causing further fluid accumulation
The net result of effusion formation is a
flattening or inversion of the diaphragm, a
mechanical dissociation of the visceral and
parietal pleura, and an eventual restrictive
ventilatory defect as measured by pulmonary
function testing.
Pleural effusions are
generally classified as transudates or exudates,
based on the mechanism of fluid formation and
pleural fluid chemistry. Transudates result from
an imbalance of oncotic and hydrostatic
pressures, whereas exudates are the result of
inflammatory processes of the pleura and/or
decreased lymphatic drainage. In some cases, it
is not rare for pleural fluid to exhibit mixed
characteristics of transudate and exudate.
Transudates
Transudates result from an
imbalance in oncotic and hydrostatic pressures.
Transudative effusions are usually
ultrafiltrates of plasma squeezed out of the
pleura as a result of an imbalance in
hydrostatic and oncotic forces in the chest.
However, other mechanisms of injury may include
upward movement of fluid from the peritoneal
cavity or, in iatrogenic cases, direct infusion
into the pleural space from misplaced (or even
migrated) central venous catheters or
nasogastric feeding tubes.
Transudates are
caused by a small, defined group of etiologies,
including the following:
• Congestive heart
failure
• Cirrhosis (hepatic hydrothorax)
• Atelectasis (may be due to occult malignancy
or pulmonary embolism)
• Hypoalbuminemia
•
Nephrotic syndrome
• Peritoneal dialysis
•
Myxedema
• Constrictive pericarditis
•
Urinothorax (usually due to obstructive
uropathy)
• Cerebrospinal fluid (CSF) leaks
to the pleura (in the setting of
ventriculopleural shunting or of trauma/surgery
to the thoracic spine)
Duropleural fistula
(rare, but may be a complication of spinal cord
surgery)
• Extravascular migration of central
venous catheter
• Glycinothorax (rare
complication of bladder irrigation with 1.5%
glycine solution following urologic surgery)
Exudates
Produced by a variety of
inflammatory conditions (and often requiring a
more extensive evaluation and treatment strategy
than transudates), exudative effusions develop
from inflammation of the pleura or from
decreased lymphatic drainage at pleural edges.
Mechanisms of exudative formation include
pleural or parenchymal inflammation, impaired
lymphatic drainage of the pleural space,
transdiaphragmatic cephalad movement of
inflammatory fluid from the peritoneal space,
altered permeability of pleural membranes,
and/or increased capillary wall permeability or
vascular disruption. Pleural membranes are
involved in the pathogenesis of the fluid
formation. Of note, the permeability of pleural
capillaries to proteins is increased in disease
states with elevated protein content.
The
more common causes of exudates include the
following:
Parapneumonic causes
Malignancy
(most commonly lung or breast cancer, lymphoma,
and leukemia; less commonly ovarian carcinoma,
stomach cancer, sarcomas, melanoma)[9]
Pulmonary embolism
Collagen-vascular
conditions (rheumatoid arthritis, systemic lupus
erythematosus)
Tuberculosis (TB)
Pancreatitis Trauma
Postcardiac injury
syndrome Esophageal perforation Radiation
pleuritis Sarcoidosis
Fungal infection
Pancreatic pseudocyst Intra-abdominal abscess
Status post coronary artery bypass graft (CABG)
surgery Pericardial disease
Meigs syndrome
(benign pelvic neoplasm with associated ascites
and pleural effusion)
Ovarian
hyperstimulation syndrome
Drug-induced
pleural disease (see Pneumotox.com for an
extensive and searchable list of drugs that may
cause pleural effusion)
Asbestos-related
pleural disease
Yellow nail syndrome (yellow
nails, lymphedema, pleural effusions)
Uremia
Trapped lung (localized pleural scarring with
the formation of a fibrin peel prevents
incomplete lung expansion, at times leading to
pleural effusion)
Chylothorax (acute illness
with elevated triglycerides in pleural fluid)
Pseudochylothorax (chronic condition with
elevated cholesterol in pleural fluid) istula
(ventriculopleural, biliopleural, gastropleural)
Epidemiology
Occurrence in the United
States
Since pleural effusion is usually the
manifestation of an underlying disease process,
its precise incidence is difficult to determine.
Nevertheless, the incidence in the United States
is estimated to be at least 1.5 million cases
annually.Most of these cases are caused by
congestive heart failure, bacterial pneumonia,
malignancy, and pulmonary embolism.
International occurrence
The estimated
prevalence of pleural effusion is 320 cases per
100,000 people in industrialized countries, with
a distribution of etiologies related to the
prevalence of underlying diseases.
Sex-related demographics
Although certain
etiologies have a sex predilection, the general
understanding is that the incidence of pleural
effusion is equal between the sexes. Nearly two
thirds of malignant pleural effusions occur in
women, in whom they are associated with breast
and gynecologic malignancies.
Pleural
effusion associated with systemic lupus
erythematosus is specifically more common in
women than in men. In the United States, the
incidence of pleural effusion in the setting of
malignant mesothelioma is higher in men,
probably because of their higher occupational
exposure to asbestos.
Pleural effusions
associated with chronic pancreatitis are more
common in men, with the majority of male cases
having alcohol abuse as the impetus. Rheumatoid
effusions also occur more commonly in males than
in females.
Race- and age-related
demographics
Since pleural effusion is
usually the manifestation of an underlying
disease process, racial differences most likely
reflect racial variation in incidence of the
causative disorder.
Pleural effusions usually
occur in adults. However, they appear to be
increasing in children, often in the setting of
underlying pneumonia. Fetal pleural effusions
have also been reported and under certain
circumstances may be treated prior to delivery.
Prognosis
The prognosis in pleural effusion
varies in accordance with the condition's
underlying etiology. However, patients who seek
medical care earlier in the course of their
disease and those who obtain prompt diagnosis
and treatment have a substantially lower rate of
complications than do patients who do not.
Morbidity and mortality
Morbidity and
mortality of pleural effusions are directly
related to cause (and if applicable, staging) of
the underlying disease at the time of
presentation, as well as biochemical findings in
the pleural fluid.
Morbidity and mortality
rates in patients with pneumonia and pleural
effusions are higher than those in patients with
pneumonia alone. Parapneumonic effusions, when
recognized and treated promptly, typically
resolve without significant sequelae. However,
untreated or inappropriately treated
parapneumonic effusions may lead to empyema,
constrictive fibrosis, and sepsis.
Development of a malignant pleural effusion is
associated with a very poor prognosis, with
median survival of 4 months and mean survival of
less than 1 year. The most common associated
malignancy in men is lung cancer. The most
common associated malignancy in women is breast
cancer. Median survival ranges from 3-12 months,
depending on the malignancy. Effusions from
cancers that are more responsive to
chemotherapy, such as lymphoma or breast cancer,
are more likely to be associated with prolonged
survival, compared with those from lung cancer
or mesothelioma.
Cellular and biochemical
findings in the fluid may also be indicators of
prognosis. For example, a lower pleural fluid pH
is often associated with a higher tumor burden
and a worse prognosis.
Presentation
History
A detailed medical
history should be obtained from all patients
presenting with a pleural effusion, as this may
help to establish the etiology. For example, a
history of chronic hepatitis or alcoholism with
cirrhosis suggests hepatic hydrothorax or
alcohol-induced pancreatitis with effusion.
Recent trauma or surgery to the thoracic spine
raises the possibility of a CSF leak. The
patient should be asked about a history of
cancer, even remote, as malignant pleural
effusions can develop many years after initial
diagnosis.
An occupational history should
also be obtained, including potential asbestos
exposure, which could predispose the patient to
mesothelioma or benign asbestos-related pleural
effusion. The patient should also be asked about
medications they are taking.
The clinical
manifestations of pleural effusion are variable
and often related to the underlying disease
process. The most commonly associated symptoms
are progressive dyspnea, cough, and pleuritic
chest pain.
Dyspnea
Dyspnea is the most
common symptom associated with pleural effusion
and is related more to distortion of the
diaphragm and chest wall during respiration than
to hypoxemia. In many patients, drainage of
pleural fluid alleviates dyspnea despite limited
alterations in gas exchange. Drainage of pleural
fluid may also allow the underlying disease to
be more easily recognized on repeat chest
radiographs. Note that dyspnea may be caused by
the condition producing the pleural effusion,
such as underlying intrinsic lung or heart
disease or obstructing endobronchial lesions
rather than by the effusion itself.
Cough
Cough in patients with pleural effusion is often
mild and nonproductive. More severe cough or the
production of purulent or bloody sputum suggests
an underlying pneumonia or endobronchial lesion.
Chest pain
The presence of chest pain, which
results from pleural irritation, raises the
likelihood of an exudative etiology, such as
pleural infection, mesothelioma, or pulmonary
infarction.
Pain may be mild or severe. It is
typically described as sharp or stabbing and is
exacerbated with deep inspiration. Pain may be
localized to the chest wall or referred to the
ipsilateral shoulder or upper abdomen due to
diaphragmatic irritation. Pain may decrease in
intensity as the pleural effusion increases in
size and the inflamed pleural surfaces are no
longer in contact with each other.
Extrapulmonary symptoms
Other symptoms in
association with pleural effusions may suggest
the underlying disease process. Increasing lower
extremity edema, orthopnea, and paroxysmal
nocturnal dyspnea may all occur with congestive
heart failure.
Night sweats, fever,
hemoptysis, and weight loss should suggest TB.
Hemoptysis also raises the possibility of
malignancy, other endotracheal or endobronchial
pathology, or pulmonary infarction. An acute
febrile episode, purulent sputum production, and
pleuritic chest pain may occur in patients with
an effusion associated with pneumonia.
Physical Examination
Physical findings in
pleural effusion are variable and depend on the
volume of the effusion. Typically, there are no
clinical findings for effusions less than 300
mL. With effusions greater than 300 mL, chest
wall/pulmonary findings may include the
following:
Dullness to percussion, decreased
tactile fremitus, and asymmetrical chest
expansion, with diminished or delayed expansion
on the side of the effusion: These are the most
reliable physical findings of pleural effusion:
/lediastinal shift away from the effusion: This
finding is observed with effusions greater than
1000 mL. Displacement of ie trachea and
mediastinum towards the side of the effusion is
an important clue to obstruction of a lobar
bronchus by an airway; bronchial lesion, which
can be due to malignancy or, less commonly, to a
nonmalignant cause, such as a foreign body
obstruction.
• Diminished or inaudible breath
sounds
• Egophony (known as "E-to-A" changes)
at the most superior aspect of the pleural
effusion
• Pleural friction rub
Other
physical and extrapulmonary findings may suggest
the underlying cause of the pleural effusion.
Peripheral edema, distended neck veins, and S3
gallop suggest congestive heart failure. Edema
may also be a manifestation of nephrotic
syndrome, pericardial disease, or, when combined
with yellow nailbeds, the yellow nail syndrome.
Cutaneous changes and ascites suggest liver
disease.
Lymphadenopathy or a palpable mass
suggests malignancy.
Diagnostic Considerations Transudative
pleural effusion
Considerations in the
differential diagnosis of transudative pleural
effusion include the following:
• Congestive
heart failure (most common)
• Cirrhosis with
hepatic hydrothorax
• Nephrotic syndrome
•
Peritoneal dialysis/continuous ambulatory
peritoneal dialysis
• Hypoproteinemia
•
Glomerulonephritis
• Superior vena cava
obstruction
• Fontan procedure
•
Urinothorax
■ CSF leak to the pleural space
Exudative pleural effusion
Conditions to
consider in the differential diagnosis of
exudative pleural effusion include the
following:
• Malignancy
• Pneumonia
•
Tuberculosis
• Pulmonary embolism
• Fungal
infection
• Pancreatic pseudocyst
•
Intra-abdominal abscess
Post CABG surgery
Postcardiac injury syndrome
• Pericardial
disease
• Meigs syndrome
• Ovarian
hyperstimulation syndrome
• Rheumatoid
pleuritis
• Lupus erythematosus
•
Drug-induced pleural disease
• Asbestos
pleural effusion
• Yellow nail syndrome
•
Uremia
• Trapped lung
• Chylothorax
•
Pseudochylothorax
• Acute respiratory
distress syndrome
• Chronic pleural
thickening
• Malignant mesothelioma
•
Hypothyroidism
Additional causes of pleural
effusion or mimics of pleural effusion are as
follows:
• Congestive heart failure and
pulmonary edema
• Diaphragmatic injuries
•
Esophageal rupture and tears
• Hypothyroidism
and myxedema coma
• Lung neoplasms
•
Pancreatitis
• Q fever
• Rheumatoid
arthritis
Workup
Approach Considerations
Thoracentesis should be performed for new and
unexplained pleural effusions when sufficient
fluid is present to allow a safe procedure.
Observation of pleural effusion is reasonable
when benign etiologies are likely, as in the
setting of overt congestive heart failure, viral
pleurisy, or recent thoracic or abdominal
surgery.
Laboratory testing helps to
distinguish pleural fluid transudates from
exudates. However, certain types of exudative
pleural effusions might be suspected simply by
observing the gross characteristics of the fluid
obtained during thoracentesis. Note the
following:
• Frankly purulent fluid indicates
an empyema • A putrid odor suggests an anaerobic
empyema milky, opalescent fluid suggests a
chylothorax, resulting most often from lymphatic
obstruction by malignancy or oracic duct injury
by trauma or surgical procedure
• Grossly
bloody fluid may result from trauma, malignancy,
post-pericardiotomy syndrome, or
asbestos-related effusion and indicates the need
for a spun hematocrit test of the sample. A
pleural fluid hematocrit level of more than 50%
of the peripheral hematocrit level defines a
hemothorax, which often requires tube
thoracostomy
• Black pleural fluid suggests a
limited number of diseases, including infection
with Aspergillus niger or Rizopus oryzae,
malignant melanoma, non-small cell lung cancer
or ruptured pancreatic pseudocyst, or
charcoal-containing empyema
Normal pleural fluid
Normal pleural fluid
has the following characteristics:
• Clear
ultrafiltrate of plasma that originates from the
parietal pleura
• A pH of 7.60-7.64
•
Protein content of less than 2% (1-2 g/dL)
•
Fewer than 1000 white blood cells (WBCs) per
cubic millimeter
• Glucose content similar to
that of plasma
• Lactate dehydrogenase (LDH)
less than 50% of plasma
Distinguishing Transudates From Exudates
The initial diagnostic consideration is
distinguishing transudates from exudates.
Although a number of chemical tests have been
proposed to differentiate pleural fluid
transudates from exudates, the tests first
proposed by Light et al have become the
criterion standards.
The fluid is considered
an exudate if any of the following are found:
• Ratio of pleural fluid to serum protein
greater than 0.5
• Ratio of pleural fluid to
serum LDH greater than 0.6
• Pleural fluid
LDH greater than two thirds of the upper limits
of normal serum value The fluid is considered a
transudate if all of the above are absent.
These criteria require simultaneous measurement
of pleural fluid and serum protein and LDH.
However, a meta-analysis of 1448 patients
suggested that the following combined pleural
fluid measurements might have sensitivity and
specificity comparable to the criteria from
Light et al for distinguishing transudates from
exudates:
• Pleural fluid LDH value greater
than 0.45 of the upper limit of normal serum
values
• Pleural fluid cholesterol level
greater than 45 mg/dL
• Pleural fluid protein
level greater than 2.9 g/dL
Clinical judgment
is required when pleural fluid test results fall
near the cutoff points.
The criteria from
Light et al and these alternative criteria
identify nearly all exudates correctly, but they
misclassify approximately 20-25% of transudates
as exudates, usually in patients on long-term
diuretic therapy for congestive heart failure
(because of the concentrating effect of diuresis
on protein and LDH levels within the pleural
space).
Using the criterion of serum minus
pleural protein concentration level of less than
3.1 g/dL, rather than a serum/pleural fluid
ratio of greater than 0.5, more correctly
identifies exudates in these patients.
Although pleural fluid albumin is not typically
measured, a gradient of serum albumin to pleural
fluid albumin of less than 1.2 g/dL also
identifies an exudate in such patients. Button,
studies suggest that pleural fluid levels of
N-terminal pro-brain natriuretic peptide
(NT-proBNP) are elevated in s due to congestive
heart failure. Moreover, elevated pleural
NT-proBNP was demonstrated to out-perform fluid
BNP as a marker of heart failure-related
effusion.[33] Thus, at institutions where this
test is available, high pleural levels of
NT-proBNP (defined in different studies as
>1300-4000 ng/L) may help to confirm heart
failure as the cause of an otherwise idiopathic
chronic effusion.
In a more recent systematic
review, pleural fluid cholesterol greater than
55 mg/dL and pleural LDH greater than 200 U/L
each had better positive and negative likelihood
ratio for distinguishing exudates from
transudates than did Light's criteria.
Pleural Fluid LDH, Glucose, and pH
Pleural
fluid LDH
Pleural fluid LDH levels greater
than 1000 IU/L suggest empyema, malignant
effusion, rheumatoid effusion, or pleural
paragonimiasis. Pleural fluid LDH levels are
also increased in effusions from Pneumocystis
jiroveci (formerly, P carinii) pneumonia. The
diagnosis is suggested by a pleural fluid/serum
LDH ratio of greater than 1, with a pleural
fluid/serum protein ratio of less than 0.5.
Pleural fluid glucose and pH
In addition to
the previously discussed tests, glucose and
pleural fluid pH should be measured during the
initial thoracentesis in most situations.
A
low pleural glucose concentration (30-50 mg/dL)
suggests malignant effusion, tuberculous
pleuritis, esophageal rupture, or lupus
pleuritis. A very low pleural glucose
concentration (ie, < 30 mg/dL) further restricts
diagnostic possibilities, to rheumatoid pleurisy
or empyema.
Pleural fluid pH is highly
correlated with pleural fluid glucose levels. A
pleural fluid pH of less than 7.30 with a normal
arterial blood pH level is caused by the same
diagnoses as listed above for low pleural fluid
glucose. However, for parapneumonic effusions, a
low pleural fluid pH level is more predictive of
complicated effusions (that require drainage)
than is a low pleural fluid glucose level. In
such cases, a pleural fluid pH of less than
7.1-7.2 indicates the need for urgent drainage
of the effusion, while a pleural fluid pH of
more than 7.3 suggests that the effusion may be
managed with systemic antibiotics alone.
In
malignant effusions, a pleural fluid pH of less
than 7.3 has been associated in some reports
with more extensive pleural involvement, higher
yield on cytology, decreased success of
pleurodesis, and shorter survival times.
Handle pleural fluid samples as carefully as
arterial samples for pH measurements, with fluid
collected in heparinized syringes and ideally
transported on ice for measurement within six
hours. However, studies have determined that
when collected in heparinized syringes, pleural
fluid pH does not change significantly even over
several hours at room temperature. Consequently,
if appropriately collected samples can be
processed quickly, pH measurements should not be
canceled simply because the sample was not
transported on ice.
Pleural Fluid Cell Count Differential
If
an exudate is suspected clinically or is
confirmed by chemistry test results, send the
pleural fluid for total and differential cell
counts, Gram stain, culture, and cytology.
Pleural fluid lymphocytosis, with lymphocyte
values greater than 85% of the total nucleated
cells, suggests TB, lymphoma, sarcoidosis,
chronic rheumatoid pleurisy, yellow nail
syndrome, and chylothorax. Pleural lymphocyte
values of 50-70% of the nucleated cells
suggest malignancy.
Pleural fluid
eosinophilia (PFE), with eosinophil values
greater than 10% of nucleated cells, is seen
in approximately 10% of pleural effusions and is
not correlated with peripheral blood
eosinophilia. PFE is most often caused by air or
blood in the pleural space. Blood in the pleural
space causing PFE may be the result of pulmonary
embolism with infarction or benign asbestos
pleural effusion. PFE may be associated with
other nonmalignant diseases, including parasitic
disease (especially paragonimiasis), fungal
infection (coccidioidomycosis, cryptococcosis,
histoplasmosis), and a variety of medications.
The presence of PFE does not exclude a malignant
effusion, especially in patient populations with
a high prevalence of malignancy. The presence of
PFE makes tuberculous pleurisy unlikely and also
makes the progression of a parapneumonic to an
empyema unlikely.
lial cells are found in
variable numbers in most effusions, but their
presence at greater than 5% of total nucleated
cells diagnosis of TB less likely. Markedly
increased numbers of mesothelial cells,
especially in bloody or eosinophilic.
Pleural Fluid Culture and Cytology
Cultures of infected pleural fluids yield
positive results in approximately 60% of cases.
This occurs even less often for anaerobic
organisms. Diagnostic yields, particularly for
anaerobic pathogens, may be increased by
directly culturing pleural fluid into blood
culture bottles.
Malignancy is suspected in
patients with known cancer or with lymphocytic,
exudative effusions, especially when bloody.
Direct tumor involvement of the pleura is
diagnosed most easily by performing pleural
fluid cytology.
Heparinized samples (1 mL of
1:1000 heparin per 50 mL of pleural fluid)
should be submitted for analysis if the pleural
fluid is bloody and they should be refrigerated
if samples will not be processed within one
hour.
The reported diagnostic yields in
cytology vary from 60-90%, depending on the
extent of pleural involvement and the type of
primary malignancy. Cytology findings are
positive in 58% of effusions related to
mesothelioma.
The sensitivity of cytology is
not highly related to the volume of pleural
fluid tested. Sending more than 50-60 mL of
pleural fluid for cytology does not increase the
yield of direct cytospin analysis, and volumes
of approximately 150 mL are sufficient when both
cytospin and cell block preparations are
analyzed.
Tumor markers, such as
carcinoembryonic antigen, Leu-1, and mucin, are
suggestive of malignant effusions (especially
adenocarcinoma) when pleural fluid values are
very high. However, because of low sensitivity,
they are not helpful if the values are normal
or only modestly increased.
Tuberculous
pleuritis
Suspect tuberculous pleuritis in
patients with a history of exposure or a
positive PPD finding and in patients with
lymphocytic exudative effusions, especially if
less than 5% mesothelial cells are detected on
differential cell counts.
Because most
tuberculous pleural effusions probably result
from a hypersensitivity reaction to the
Mycobacterium rather than from microbial
invasion of the pleura, acid-fast bacillus
stains of pleural fluid are rarely diagnostic (<
10% of cases). Pleural fluid cultures grow M
tuberculosis in less than 65% of cases.
In
contrast, the combination of histology and
culture of pleural tissue obtained by pleural
biopsy increases the diagnostic yield for TB to
90%.
Adenosine deaminase (ADA) activity of
greater than 43 U/mL in pleural fluid supports
the diagnosis of tuberculous pleuritis. However,
the test has a sensitivity of only 78%.
Therefore, pleural ADA values of less than
43-50 U/mL do not exclude the diagnosis of TB
pleuritis.
Interferon-gamma concentrations of
greater than 140 pg/mL in pleural fluid also
support the diagnosis of tuberculous pleuritis.
Unfortunately, this test is not routinely
available.
Additional Laboratory Tests
Additional specialized tests are warranted when
specific etiologies are suspected. Measure
pleural fluid amylase levels if a pancreatic
origin or ruptured esophagus is suspected or if
a unilateral, left-sided pleural effusion
remains undiagnosed after initial testing. Of
note, increased pleural fluid amylase can also
be seen with malignancy. An additional assay of
amylase isoenzymes can help distinguish a
pancreatic source (diagnosed by elevated pleural
fluid pancreatic isoenzymes) from other
etiologies.
Measure triglyceride and
cholesterol levels in milky pleural fluids when
chylothorax or pseudochylothorax is suspected.
Consider immunologic studies, including pleural
fluid antinuclear antibody and rheumatoid
factor, when collagen-vascular diseases are
suspected.
Scanning and Ultrasonography
A study
involving 41 consecutive patients with hepatic
hydrothorax indicated that hepatic hydrothorax
virtually always presents with ascites that can
be revealed by ultrasonography or computed
tomography (CT) scanning. Point of care bedside
ultrasonography has become the standard of care
in many facilities. Also see Pleural Effusion
Imaging.
Chest CT scanning with contrast
should be performed in all patients with an
undiagnosed pleural effusion, if it has not
previously been performed, to detect thickened
pleura or signs of invasion of underlying or
adjacent structures. The two diagnostic
imperatives in this situation are pulmonary
embolism and tuberculous pleuritis. In both
cases, the pleural effusion is a harbinger of
potential future morbidity. In contrast, a short
delay in diagnosing metastatic malignancy to the
pleural space has less impact on future clinical
outcomes. CT angiography should be ordered if
pulmonary embolism is strongly suggested. Also
see Pleural Effusion Imaging.
Chest Radiography
Effusions of more than
175 mL are usually apparent as blunting of the
costophrenic angle on upright posteroanterior
chest radiographs. On supine chest radiographs,
which are commonly used in the intensive care
setting, moderate to large pleural effusions may
appear as a homogeneous increase in density
spread over the lower lung fields. Apparent
elevation of the hemidiaphragm, lateral
displacement of the dome of the diaphragm, or
increased distance between the apparent left
hemidiaphragm and the gastric air bubble
suggests subpulmonic effusions.
Bilateral
pleural effusions with loss of bilateral
costophrenic sulci (meniscus sign).
Anteroposterior, upright chest radiograph.
Lateral decubitus films more reliably detect
smaller pleural effusions. Layering of an
effusion on lateral decubitus films defines a
freely flowing effusion and, if the layering
fluid is 1 cm thick, indicates an effusion of
greater than 200 mL that is amenable to
thoracentesis. Failure of an effusion to layer
on lateral decubitus films indicates the
presence of loculated pleural fluid or some
other etiology causing the increased pleural
density. Note that decubitus films are almost
never performed in those institutions with
bedside ultrasonography.
Left lateral
decubitus film displaying freely layering
left-sided pleural effusion.
Idiopathic Exudative Effusions
Despite
evaluations with repeated diagnostic
thoracenteses, approximately 20% of exudative
effusions remain undiagnosed. Clues to the
diagnosis that may have been overlooked include
(1) occupational exposure to asbestos 10-20
years earlier, which may suggest benign asbestos
effusion; (2) medication exposure to
nitrofurantoin, amiodarone, or medications
associated with a drug-induced lupus syndrome;
and (3) hepatic hydrothorax unrecognized in a
patient with minimal or undetectable ascites.
Among patients with undiagnosed pleural
effusions after the primary evaluation, those
who meet all 6 of the following clinical
parameters are predicted to have a benign
course, and no further evaluation is necessary:
• Patients are clinically stable
• Patients
do not have weight loss
• The results of the
purified protein derivative (PPD) test, used in
detecting tuberculous pleural effusion, are
negative and the pleural adenosine deaminase
(ADA) value, also used in diagnosing tuberculous
pleural effusion, is less than 43 U/mL
• The
patient does not have a fever
• The pleural
fluid differential blood cell count has less
than 95% lymphocytes
• The effusion occupies
less than 50% of the hemithorax
For other
patients with undiagnosed exudative effusions,
approximately 20% have a specific etiology
determined, including malignancy. For such
patients, weigh the benefits and risks of
pursuing a diagnostic strategy that will involve
using progressively more invasive procedures,
given the low likelihood of finding a curable
etiology. Note the following:
• Bronchoscopy
- Consider only if a patient has parenchymal
abnormalities or hemoptysis
• Surgical
approaches to the diagnosis of pleural effusions
- Includes video-assisted thoracoscopy
(pleuroscopy) and open thoracotomy, allows
direct visualization and biopsy of the pleura
for diagnosis of exudative effusions, which
reveals an etiology in 92% of effusions that
remain undiagnosed after a medical evaluation,
with an operative mortality of less than 0.5%
• Medical thoracoscopy - Where available, may be
diagnostic and therapeutic; complete drainage of
the effusion and talc sclerosis can be performed
at the time of the procedure
Note that in
most medical centers, surgical exploration using
thoracoscopy or thoracotomy entails the risks of
general anesthesia and is probably warranted
only in patients who are symptomatic and anxious
for a (potentially incurable) diagnosis.
Please see also our Toxilact data base which is in the following language versions:
Toxilact Deutsche Sprachversion
Toxilact Nederlandstalige versie
Toxilakt έκδοση στην ελληνική γλώσσα
Toxilact English language version
Toxilact magyar nyelvű változat
Toxilact versione in lingua italiana
Toxilact polska wersja językowa
Pulmonary symptoms, findings and investigations
Assessment of chronic cough
Pleural Effusion
Approach to wheezing in children
Polysomnography
Pulmonary Function Testing
Toxicological risk during lactation
Toxicological lactation category I - the drug and/or its metabolites are either not eliminated through breast milk or are not toxic to the newborn and cannot lead to the development of absolutely any toxic reactions and adverse consequences for his health in the near and long term. Breast-feeding does not need to be discontinued while taking a given drug that falls into this toxicological lactation category.
Toxicological lactation category II - the drug and its metabolites are also eliminated through breast milk, but the plasma:milk ratio is very low and/or the excreted amounts cannot generate toxic reactions in the newborn due to various reasons, including degradation of the drug in the acid pool of the stomach of the newborn. Breastfeeding does not need to be discontinued while taking this medicine.
Toxicological lactation category III - the drug and/or its metabolites generate in breast milk equal to plasma concentrations or higher, and therefore the possible development of toxic reactions in the newborn can be expected. Breastfeeding should be discontinued for the period corresponding to the complete elimination of the drug or its metabolites from the mother's plasma.
Toxicological lactation category IV - the drug and/or its metabolites generate a plasma:milk ratio of 1:1 or higher and/or have a highly toxic profile for both the mother and the newborn, therefore their administration is incompatible with breastfeeding and it should to stop completely, and not just for the period of taking the drug, or to look for a less toxic therapeutic alternative.